Sulfation (alias sulfurylation) of bioactive compounds (e.g. flavonoids and phenolic acids), which are part of a (healthy) human diet, is an important reaction in nature as it plays a significant role in their detoxication via Phase II metabolism. Due to the low bioavailability of the parent compounds, their metabolites are likely responsible for their biological activity. Therefore, sulfated compounds are of interest as standards for metabolic studies and for the evaluation of their bioactivity. Aryl-sulfotransferases can serve as an efficient biosynthetic tool for the preparation of such sulfated metabolites.
Mammalian sulfotransferases
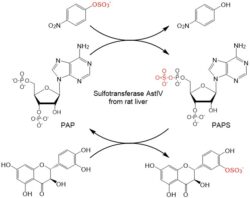
Nature has developed efficient sulfation tools using 3′-phosphoadenosine 5′-phosphosulfate (PAPS) as a sulfate donor for a variety sulfotransferases (SULTs, EC 2.8.2), which catalyze most sulfation reactions in vivo. However, these typically intracellular enzymes are inherently unstable under in vitro conditions and the sulfate donor is extremely expensive. This bottleneck can be overcome using biotransformation with whole cells expressing the respective enzymes. We have used aryl sulfotransferase IV from rat liver overexpressed in Escherichia coli for the sulfation of silybin (Purchartová 2013) and quercetin and its derivatives (Purchartová 2015).
Sulfation of (+)-taxifolin and PAPS regeneration (both) catalyzed by sulfotransferase AstIV from rat liver.
Bacterial sulfotransferases
Bacterial PAPS-independent aryl-sulfotransferases (ASTs, EC 2.8.2.1) use simple aromatic sulfate donors such as p-nitrophenyl sulfate (p-NPS). These periplasmic enzymes are quite robust, stable and offer a priceworthy alternative to synthetic applications. At present, only the enzyme from Desulfitobacterium hafniense (DhAST) and a few other ASTs are described in the literature. We have used the crude DhAST for the sulfation of silymarin flavonolignans (Marhol 2013, Valentová 2018), quercetin and related flavonoids (Purchartová 2015, Valentová 2017, Káňová 2021), simple phenols and phenolic acids including those from olive oil (Begines 2019, Petrásková 2022, Brodsky 2022).
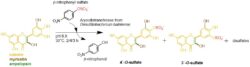
Sulfation of flavonoids with aryl-sulfotransferase from Desulfitobacterium hafniense
We have also prepared a new His-tagged construct of DhAST (DhAST-tag), purified it to homogeneity and biochemically characterized it (Brodsky 2022).


 Prof. RNDr. Pavla Bojarová, Ph.D.
Prof. RNDr. Pavla Bojarová, Ph.D.