L. Martínková, M. Chmátal, V. Křen, L.Rucká, M.Pátek, J.Nešvera
Cyanide-containing wastes are produced by chemical, mining and coal industries and must be detoxified prior to their disposal. To this end, chemical oxidation has been used, which, however, needs large amounts of harmful chemicals and may give toxic by-products. The use of microbial cultures is environmentally benign but the organisms are sensitive to higher cyanide concentrations. The use of enzymes seems to be able to solve these problems.
Microbial enzymes (cyanide dihydratases, cyanide hydratases; Figure 1) are related to nitrilases (see „Biotransformations by enzymes of the nitrile metabolism“). They are able to hydrolyze cyanide into non-toxic or less toxic products (formamide, formic acid, ammonium ions), which can be further utilized by microbes.
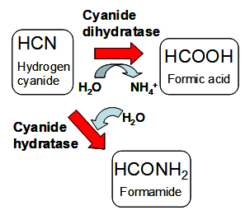
Figure 1: Detoxication of cyanide by microbial enzymes
The cyanide hydratase from Aspergillus niger is very efficient in cyanide degradation. We have produced large amounts of this enzyme in Escherichia coli and used the whole cells or the isolated enzyme to degrade high concentrations of cyanide (up to 0.65 g L-1). The enzyme retains its activity in highly polluted wastewaters such as coking effluents. The enzymatic removal of cyanide enables enzymes such as tyrosinase or laccase (cyanide sensitive) to degrade toxic phenolics in these wastewaters.
Literature
- Rinágelová A, Kaplan O, Veselá AB, Chmátal M, Křenková A, Plíhal O, Pasquarelli F, Cantarella M, Martínková L. (2014) Cyanide hydratase from Aspergillus niger K10: Overproduction in Escherichia coli, purification, characterization and use in continuous cyanide degradation. J Ind Microbiol Biotechnol 39:1811–1819
- Martínková L, Veselá AB, Rinágelová A, Chmátal M (2015) Cyanide hydratases and cyanide dihydratases: emerging tools in the biodegradation and biodetection of cyanide. Appl Microbiol Biotechnol 99:8875–8882
- Martínková L, Chmátal M (2016) The integration of cyanide hydratase and tyrosinase catalysts enables effective degradation of cyanide and phenol in coking wastewaters. Wat Res 102:90–95
Cooperation
- Abitec s.r.o., Prague, Czech Republic
- Třinec Iron and Steel Works, Třinec, Czech Republic
- M. Cantarella, University of L´Aquila, Italy


 Prof. RNDr. Pavla Bojarová, Ph.D.
Prof. RNDr. Pavla Bojarová, Ph.D.